The intestinal host-environment interface comprises one of the most complex ecosystems on Earth. Dietary nutrients as well as bacterial, viral, and eukaryotic microbiomes influences many aspects of host physiology, but at the same time needs to be tightly regulated by the intestinal epithelium. We are particularly interested in understanding the consequences of this interface on disease risk in the context of cancer, chronic inflammatory diseases, metabolic diseases, and post-infection syndromes. We study this interplay using three complementary fields of research.
Research
Intestinal epithelium
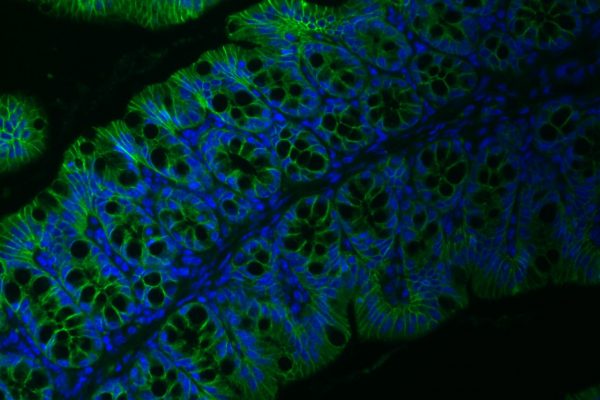
We are exploring new functions of the intestinal epithelium in the maintenance of host-diet-microbiota mutualism. A particular focus is on the integration of cellular function with the regulation of nutrient absorption and intestinal colonization by bacteria, viruses, and eukaryotes.
Metabolites

We are investigating the role of dietary and microbiome-derived metabolites as regulators of host immunity and metabolism. Of particular interest is the role of metabolites in inflammatory, infectious, and neoplastic disease.
Innate immunity
We are studying new modalities of innate immunity in classical and non-classical immune cells. We aim to decipher the integration of immune signaling pathways into cellular function.